A demonstrated long-term safety profile: Koselugo has been studied in patients with NF1 PN for up to 7.7 years1*
Duration of exposure with Koselugo
At the DCO of June 2018 in the SPRINT Phase 2 Stratum 1 study,
of patients were exposed to Koselugo for ≥12 months2
of patients were exposed to Koselugo for ≥2 years2
Known selumetinib-related adverse reactions can develop years after treatment initiation, and therefore ongoing monitoring is needed while patients remain on Koselugo.1
Adverse reactions (≥20%) in patients who received Koselugo in SPRINT Phase 2 Stratum 12†
Koselugo N=50
Gastrointestinal
Vomiting
Abdominal pain§
Diarrhea
Nausea
Stomatitis‖
Constipation
Skin and subcutaneous tissue
Rash (all)¶
Dry skin
Rash acneiform#**
Paronychia**
Pruritus
Dermatitis††
Hair changes‡‡
Musculoskeletal and connective tissue
Musculoskeletal pain§§
General
Fatigue‖‖
Pyrexia
Edema¶¶
Nervous system
Headache
Respiratory, thoracic, and mediastinal
Epistaxis
Renal and urinary system
Hematuria
Proteinuria
Metabolism and nutrition
Decreased appetite
Cardiac system
Decreased ejection fraction
Sinus tachycardia
Infections
Skin infection##
*This statement reflects exposure to Koselugo in 74 pediatric patients who received a dosage ranging from 20 mg/m² to 30 mg/m² orally twice daily in SPRINT. At the DCO of February 2021, the median duration of exposure with Koselugo was 4.4 years (range: 28 days to 7.7 years).1
†DCO June 2018.2
‡No AEs greater than Grade 3 were reported.2
§Abdominal pain includes abdominal pain, abdominal pain upper.
‖Stomatitis includes stomatitis, mouth ulceration.
¶Rash (all) includes dermatitis acneiform, rash maculopapular, erythema, rash pustular, rash, urticaria, exfoliative rash, rash pruritic, rash erythematous.
#Rash (acneiform) includes dermatitis acneiform.
**Paronychia includes paronychia, nail infection.
††Dermatitis includes dermatitis, dermatitis atopic, dermatitis diaper, eczema, seborrheic dermatitis, skin irritation
‡‡Hair changes include alopecia, hair color change.
§§Musculoskeletal pain includes pain in extremity, back pain, neck pain, musculoskeletal pain.
‖‖Fatigue includes fatigue, malaise.
¶¶Edema includes peripheral swelling, edema, localized edema.
##Skin infection includes skin infection, abscess, cellulitis, impetigo, staphylococcal skin infection.
AE=adverse event; DCO=data cutoff; NF1=neurofibromatosis type 1; PN=plexiform neurofibromas.
Dosing & AE Management Guide
A guide that provides dosing information and presents how specific toxicities were managed in the SPRINT study, based on institutional policies and guidelines
Adverse reactions are well characterized, can be manageable, and may not require discontinuation2
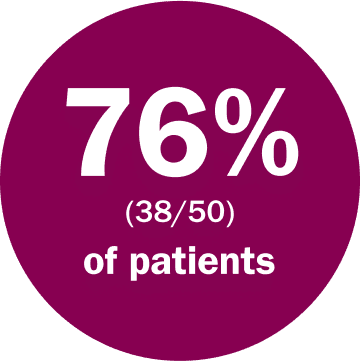
were able to stay on a full dose of Koselugo, without the need for a dose reduction***
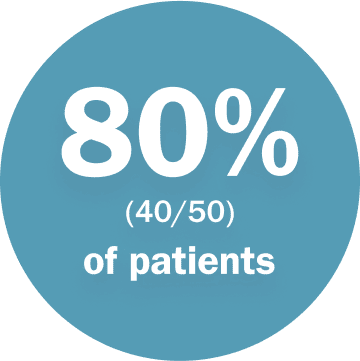
required a dose interruption but avoided discontinuation***
Adverse reactions requiring a dosage interruption or reduction in ≥5% of patients were vomiting, paronychia, diarrhea, nausea, abdominal pain, rash, skin infection, influenza-like illness, pyrexia, and weight gain.2
12% (6/50) of patients permanently discontinued due to an AE2
- These AEs included increased blood creatinine, increased weight, diarrhea, paronychia, malignant peripheral nerve sheath tumor, acute kidney injury, and skin ulcer2
- 10% (5/50) of patients discontinued due to treatment-related AEs3
- The median time to discontinuation for those 5 patients was 277 days (range: 64 days to 636 days)3
Select laboratory abnormalities (≥15%) worsening from baseline in patients who received Koselugo in SPRINT Phase 2 Stratum 12
Koselugo
Chemistry
Increased CPK
Decreased albumin
Increased AST
Increased ALT
Increased lipase
Increased potassium
Decreased potassium
Increased alkaline phosphatase
Increased amylase
Increased sodium
Decreased sodium
Hematology
Decreased hemoglobin
Decreased neutrophils
Decreased lymphocytes
In the long-term follow-up, the median duration of exposure with Koselugo was 4.4 years. During this time, no new safety signals were identified.1*§§§
***Due to adverse reactions. DCO June 2018.2
†††The denominator used to calculate the rate varied from 39 to 49 based on the number of patients with a baseline value and at least one posttreatment value.2
‡‡‡Includes one Grade 4 increased CPK and one Grade 4 increased potassium.2
§§§No new safety signals were identified compared to the earlier publications.
ALT=alanine aminotransferase; AST=aspartate aminotransferase; CPK=creatine phosphokinase.


