Koselugo allows you to prescribe with the confidence of easy-to-maintain, continuous dosing1
Recommended dosage
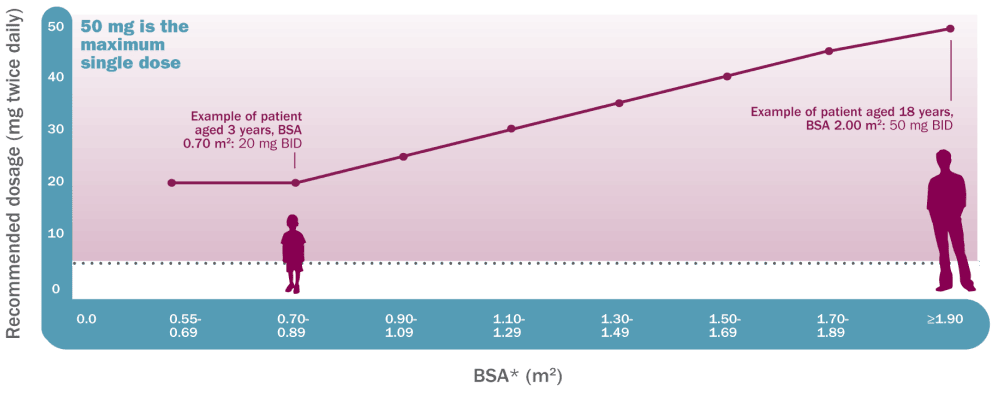
25 mg/m2 | twice daily
Dosing is individualized based on BSA (mg/
m2) and is rounded to the nearest achievable 5-mg
or 10-mg dose (up to a maximum single dose of 50 mg).

It is important to monitor your patients and reassess dosage based on their BSA changes.
Koselugo is available in 25-mg and 10-mg capsules and 28- and 60-count bottles

Recommended administration of Koselugo:
- Before prescribing, children should be assessed for the ability to swallow capsules
- Orally twice daily (approximately every 12 hours) until disease progression
-
No fasting requirement
- Swallowed whole with water: Do not chew, dissolve, or open capsule
Advise patients:
Do not take a missed dose unless it is more than 6 hours until the next scheduled dose
If vomiting occurs, do not take an additional dose but continue with the next scheduled dose
Testing requirements1
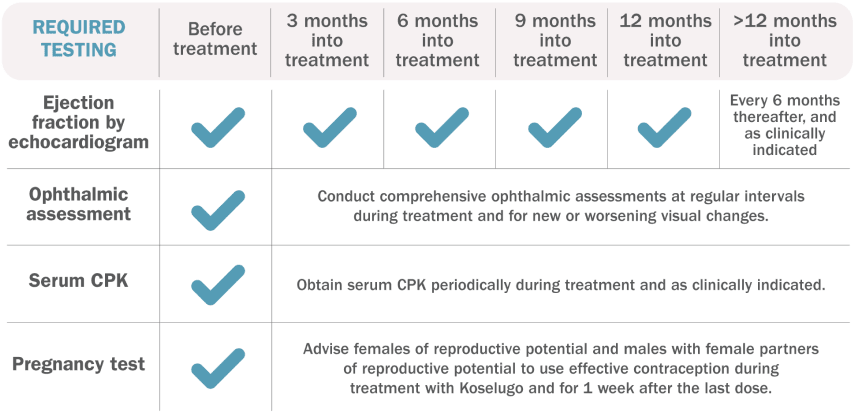
Additional Evaluation Guidelines1:
Ejection Fraction by Echocardiogram: Withhold, reduce dose, or permanently discontinue Koselugo based on severity of adverse reaction. In patients who interrupt Koselugo for decreased LVEF, obtain an echocardiogram or a cardiac MRI every 3 to 6 weeks. Upon resolution of decreased LVEF to greater than or equal to the institutional LLN, obtain an echocardiogram or a cardiac MRI every 2 to 3 months or as directed by the cardiologist.
Ophthalmic Assessment: Permanently discontinue Koselugo in patients with RVO. Withhold Koselugo in patients with RPED, follow up with optical coherence tomography assessments every 3 weeks until resolution, and resume Koselugo at a reduced dose. For other ocular toxicities, withhold, reduce dose, or permanently discontinue Koselugo based on severity of adverse reaction.
Serum CPK: If increased CPK occurs, evaluate patients for rhabdomyolysis or other causes. Withhold, reduce dose, or permanently discontinue Koselugo based on severity of adverse reaction.
Pregnancy Test: Assess the pregnancy status of females of reproductive age. Advise pregnant women of the potential risk to a fetus.
BSA=body surface area; CPK=creatine phosphokinase; LLN=lower limit of normal; LVEF=left ventricular ejection fraction; MRI=magnetic resonance imaging; RPED=retinal pigment epithelial detachment; RVO=retinal vein occlusion.
Koselugo dosing is based on BSA1
Recommended dosage based on BSA
Recommended dosage and dose reductions for adverse reactions on Koselugo
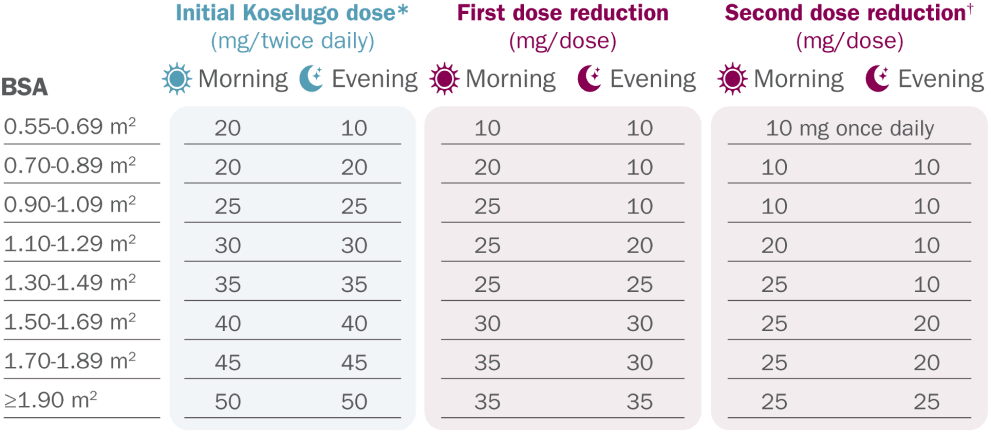
*The recommended dosage for patients with a BSA less than 0.55 m2 has not been established.
†Permanently discontinue Koselugo in patients unable to tolerate Koselugo after 2 dose reductions.
BSA=body surface area; BID=twice daily.
Hepatic Impairment‡
Drug Interactions‡
‡For recommended dosage in hepatic impairment and dosage modifications due to drug interactions, see Tables 4 and 5 in the Prescribing Information.
Dosage modifications1
Recommended dosage modifications for adverse reactions on Koselugo§
Severity of adverse reaction
Recommended dosage modifications for Koselugo
Cardiomyopathy
-
Asymptomatic decrease in LVEF of 10% or greater
from baseline
and less than LLN
Withhold until resolution.
Resume at reduced dose.
- Symptomatic decreased LVEF
- Grade 3 or 4 decreased LVEF
Permanently discontinue.
Ocular toxicity
- RPED
Withhold until resolution. Resume at reduced dose.
- RVO
Permanently discontinue.
Gastrointestinal toxicity
- Grade 3 diarrhea
Withhold until improved to Grade 0 or 1.
Resume at same dose.
Permanently
discontinue if no improvement within 3 days.
- Grade 4 diarrhea
Permanently discontinue.
- Grade 3 or 4 colitis
Permanently discontinue.
Skin toxicity
- Grade 3 or 4
Withhold until improvement.
Resume at
reduced dose.
Increased CPK
- Grade 4 increased CPK
-
Any increased
CPK and myalgia
Withhold until improved to Grade 0 or 1.
Resume at reduced dose. Permanently
discontinue if
no improvement within 3 weeks.
- Rhabdomyolysis
Permanently discontinue.
Other adverse reactions
- Intolerable Grade 2
- Grade 3
Withhold until improved to Grade 0 or 1.
Resume at reduced dose.
- Grade 4
Withhold until improved to Grade 0 or 1.
Resume at reduced dose. Consider discontinuation.
§Per National Cancer Institute Common Terminology Criteria for Adverse Events version 4.03.


